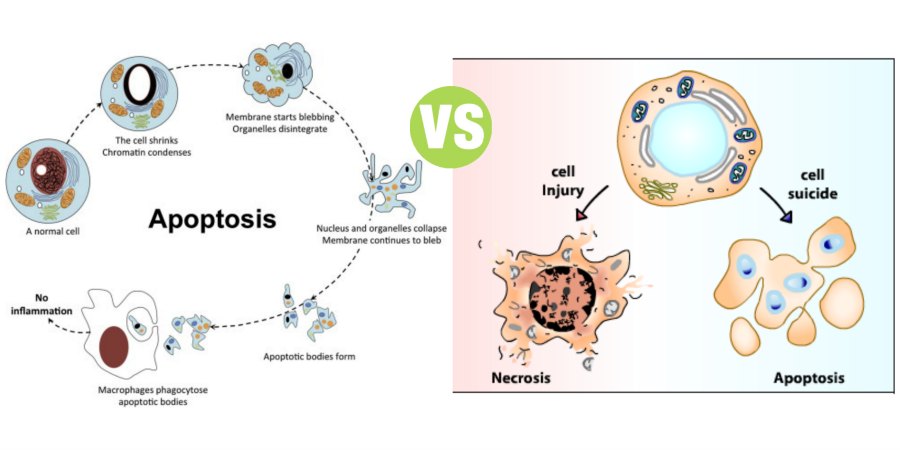
Apoptosis vs Necrosis
Summary: Difference between Apoptosis and Necrosis is that Apoptosis is defined as the natural or programmed death of the cell under genetic control. Originally, apoptosis refers to the process by which the leaves fall from trees in autumn (In Greek, apoptosis means ‘falling leaves’). While Necrosis (means ‘dead’ in Greek) is the uncontrolled and unprogrammed death of cells due to unexpected and accidental damage. It is also called ‘cell murder’ because the cell is killed by extracellular or external events.
Apoptosis
Apoptosis is defined as the natural or programmed death of the cell under genetic control. Originally, apoptosis refers to the process by which the leaves fall from trees in autumn (In Greek, apoptosis means ‘falling leaves’). It is also called ‘cell suicide’ since the genes of the cell play a major role in the death. This type of programmed cell death is a normal phenomenon and it is essential for normal development of the body. In contrast to necrosis, apoptosis usually does not produce inflammatory reactions in the neighboring tissues.
Functional Significance of Apoptosis
The purpose of apoptosis is to remove unwanted cells without causing any stress or damage to the neighboring cells. The functional significance of apoptosis:
- Plays a vital role in cellular homeostasis. About 10 million cells are produced everyday in human body by mitosis. An equal number of cells die by apoptosis. This helps in cellular homeostasis
- Useful for removal of a cell that is damaged beyond repair by a virus or a toxin
- An essential event during the development and in adult stage.
Examples
- A large number of neurons are produced during the development of central nervous system. But up to 50% of the neurons are removed by apoptosis during the formation of synapses between neurons
- Apoptosis is responsible for the removal of tissues of webs between fingers and toes during developmental stage in fetus
- It is necessary for regression and disappearance of duct systems during sex differentiation in fetus
- The cell that looses the contact with neighboring cells or basal lamina in the epithelial tissue dies by apoptosis. This is essential for the death of old enterocytes that shed into the lumen of intestinal glands
- It plays an important role in the cyclic sloughing of the inner layer of endometrium, resulting in menstruation
- Apoptosis removes the autoaggressive T cells and prevents autoimmune diseases.
Activation of Apoptosis
Apoptosis is activated by either withdrawal of positive signals (survival factors) or arrival of negative signals.
Withdrawal of Positive Signals
Positive signals are the signals which are necessary for the long-time survival of most of the cells. The positive signals are continuously produced by other cells or some chemical stimulants. Best examples of chemical stimulants are:
- Nerve growth factors (for neurons)
- Interleukin-2 (for cells like lymphocytes).
The absence or withdrawal of the positive signals activates apoptosis.
Arrival of Negative Signals
Negative signals are the external or internal stimuli which initiate apoptosis. The negative signals are produced during various events like:
- Normal developmental procedures
- Cellular stress
- Increase in the concentration of intracellular oxidants
- Viral infection
- Damage of DNA
- Exposure to agents like chemotherapeutic drugs, X-rays, ultraviolet rays and the death-receptor ligands.
Death-Receptor Ligands and Death Receptors
Death-Receptor Ligands are the substances which bind with specific cell membrane receptors and initiate the process of apoptosis. The common death-receptor ligands are tumor necrosis factors (TNF α, TNF β) and Fas ligand (which binds to the receptor called Fas). Deathreceptors are the cell membrane receptors which receive the death-receptor ligands. Well-characterized death receptors are TNF receptor-1 (TNFR1) and TNF-related apoptosis inducing ligand (TRAIL) receptors called DR4 and DR5.
Role of Mitochondria in Apoptosis
External or internal stimuli initiate apoptosis by activating the proteases called caspases (cysteinyl-dependent aspartatespecific proteases). Normally, caspases are suppressed by the inhibitor protein called apoptosis inhibiting factor (AIF).
When the cells receive the apoptotic stimulus, mitochondria releases two protein materials. First one is Cytochrome C and the second protein is called second mitochondria-derived activator of caspases (SMAC) or its homologudiablo.
SMAC/diablo inactivates AIF so that the inhibitor is inhibited. During this process, SMAC/diablo and AIF aggregate to form apoptosome which activates caspases. Cytochrome C also facilitates caspase activation.
Apoptotic Process
Cell shows sequence of characteristic morphological changes during apoptosis, viz.:
- Activated caspases digest the proteins of cytoskeleton and the cell shrinks and becomes round
- Because of shrinkage, the cell losses the contact with neighboring cells or surrounding matrix
- Chromatin in the nucleus undergoes degradation and condensation
- Nuclear membrane becomes discontinuous and the DNA inside nucleus is cleaved into small fragments
- Following the degradation of DNA, the nucleus breaks into many discrete nucleosomal units, which are also called chromatin bodies
- Cell membrane breaks and shows bubbled appearance
- Finally, the cell breaks into several fragments containing intracellular materials including chromatin bodies and organelles of the cell. Such cellular fragments are called vesicles or apoptotic bodies
- Apoptotic bodies are engulfed by phagocytes and dendritic cells.
Abnormal Apoptosis
Apoptosis within normal limits is beneficial for the body. However, too much or too little apoptosis leads to abnormal conditions.
Common abnormalities due to too much apoptosis:
- Ischemicrelated injuries
- Autoimmune diseases like:
- Hemolytic anemia
- Thrombocytopenia
- Acquired immunodeficiency syndrome (AIDS)
3. Neurodegenerative diseases like Alzheimer’s disease.
Common abnormalities due to too little apoptosis:
- Cancer
- Autoimmune lymphoproliferative syndrome (ALPS).
More Confusing Terms
Difference Between Dementia and Alzheimer’s
What is the Difference Between Mitosis and Meiosis
Difference Between Sexual and Asexual Reproduction
Difference Between Lysosomes and Peroxisomes
Difference Between Cell and Tissue
Difference Between Atrophy and Hypertrophy
NECROSIS
Necrosis (means ‘dead’ in Greek) is the uncontrolled and unprogrammed death of cells due to unexpected and accidental damage. It is also called ‘cell murder’ because the cell is killed by extracellular or external events. After necrosis, the harmful chemical substances released from the dead cells cause damage and inflammation of neighboring tissues.
Causes for Necrosis
Common causes of necrosis are injury, infection, inflammation, infarction and cancer. Necrosis is induced by both physical and chemical events such as heat, radiation, trauma, hypoxia due to lack of blood flow and exposure to toxins.
Necrotic Process
Necrosis results in lethal disruption of cell structure and activity. The cell undergoes a series of characteristic changes during necrotic process, viz.
- Cell swells causing damage of the cell membrane and appearance of many holes in the membrane
- Intracellular contents leak out into the surrounding environment
- Intracellular environment is altered
- Simultaneously, large amount of calcium ions are released by the damaged mitochondria and other organelles
- Presence of calcium ions drastically affects the organization and activities of proteins in the intracellular components
- Calcium ions also induce release of toxic materials that activate the lysosomal enzymes
- Lysosomal enzymes cause degradation of cellular components and the cell is totally disassembled resulting in death
- Products broken down from the disassembled cell are ingested by neighboring cells.
Reaction of Neighboring Tissues after Necrosis
Tissues surrounding the necrotic cells react to the breakdown products of the dead cells, particularly the derivatives of membrane phospholipids like the arachidonic acid. Along with other materials, arachidonic acid causes the following inflammatory reactions in the surrounding tissues:
- Dilatation of capillaries in the region and thereby increasing local blood flow
- Increase in the temperature leading to reddening of the tissues
- Release of histamine from these tissues which induces pain in the affected area
- Migration of leukocytes and macrophages from blood to the affected area because of increased capillary permeability
- Movement of water from blood into the tissues causing local edema
- Engulfing and digestion of cellular debris and foreign materials like bacteria by the leukocytes and macrophages
- Activation of immune system resulting in the removal of foreign materials
- Formation of pus by the dead leukocytes during this process
- Finally, tissue growth in the area and wound healing.
Also Read:
What is the Difference Between Dementia and Alzheimer’s
Difference Between DNA and RNA
Difference Between Sex and Gender



Leave a Comment
You must be logged in to post a comment.