Acidosis vs Alkalosis
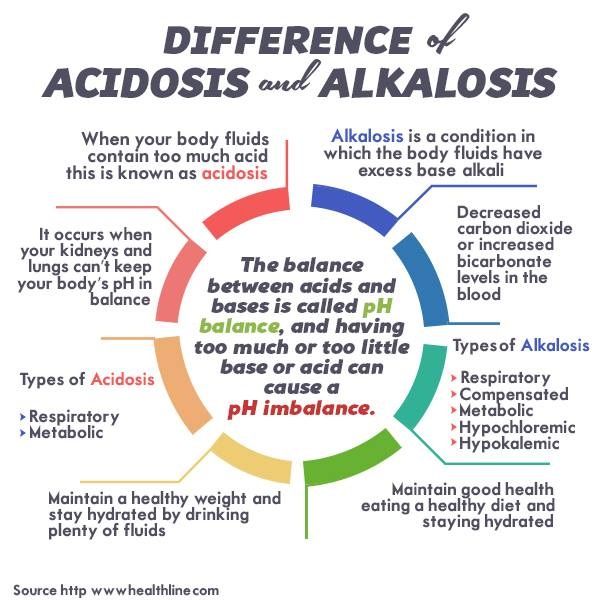
Summary: Difference Between Acidosis and Alkalosis is that Acidosis is the reduction in pH (increase in H+ concentration) below normal range. While Alkalosis is the increase in pH (decrease in H+ concentration) above the normal range.
Acidosis
Acidosis is the reduction in pH (increase in H+ concentration) below normal range. Acidosis is produced by:
- Increase in partial pressure of CO2 in the body fluids particularly in arterial blood
- Decrease in HCO3 – concentration.
Alkalosis
Alkalosis is the increase in pH (decrease in H+ concentration) above the normal range. Alkalosis is produced by:
- Decrease in partial pressure of CO2 in the arterial blood
- Increase in HCO3 – concentration.
Since the partial pressure of CO2 (pCO2 ) in arterial blood is controlled by lungs, the acid-base disturbances produced by the change in arterial pCO2 are called the respiratory disturbances.
On the other hand, the disturbances in acid-base status produced by the change in HCO3 – concentration are generally called the metabolic disturbances. Thus the acid-base disturbances are:
- Respiratory acidosis
- Respiratory alkalosis
- Metabolic acidosis
- Metabolic alkalosis.
Respiratory Acidosis
Respiratory acidosis is the acidosis that is caused by alveolar hypoventilation. During hypoventilation the lungs fail to expel CO2 , which is produced in the tissues. CO2 is the major end product of oxidation of carbohydrates, proteins and fats. CO2 accumulates in blood where it reacts with water to form carbonic acid, which is called respiratory acid. Carbonic acid dissociates into H+ and HCO3 – . The increased H+ concentration in blood leads to decrease in pH and acidosis.
Normal partial pressure of CO2 in arterial blood is about 40 mm Hg. When it increases above 60 mm Hg acidosis occurs.
Respiratory Alkalosis
Respiratory alkalosis is the alkalosis that is caused by alveolar hyperventilation. Hyperventilation causes excess loss of CO2 from the body. Loss of CO2 leads to decreased formation of carbonic acid and decreased release of H+. Decreased H+ concentration increases the pH leading to respiratory alkalosis. When the partial pressure of CO2 in arterial blood decreases below 20 mm Hg, alkalosis occurs.
Metabolic Acidosis
Metabolic acidosis is the acid-base imbalance characterized by excess accumulation of organic acids in the body, which is caused by abnormal metabolic processes. Organic acids such as lactic acid, ketoacids and uric acid are formed by normal metabolism. The quantity of these acids increases due to abnormality in the metabolism.
Metabolic Alkalosis
Metabolic alkalosis is the acid-base imbalance caused by loss of excess H+ resulting in increased HCO3 – concentration. Some of the endocrine disorders, renal tubular disorders, etc. cause metabolic disorders leading to loss of H+. It increases HCO3 – and pH in the body leading to metabolic alkalosis.
More Confusing Differences in Biology:
Difference Between Metabolic Acidosis and Metabolic Alkalosis
Difference Between Respiratory Acidosis and Respiratory Alkalosis



Leave a Comment
You must be logged in to post a comment.